The Increasingly Important Use of Rat Models in Genetic Experiments
Research scientists worldwide agree that the use of rat models for disease research and experimentation has become more relevant in the past few years. While species such as saccharomyces cerevisiae, fruit flies, and zebrafish have been used for the study of cancer, organ development, recombination or large scale mutations, the rat model offers insights into human disease that the study of other species will not allow. When animals such as mice or rats are selected as models for genetic experimentation, the main reason is the similarity of their genetic makeup to human DNA. In the case of rats specifically, their particular DNA and physiology has allowed scientists to explore a variety of physiological and pathophysiological mechanisms that would not be possible in a mouse model.

### The Rat as a Model System
Rats were the first mammalian species specifically domesticated to be used in the laboratory. Two centuries ago, the brown rat Rattus norvegicus was used by scientists to understand human physiology and medicine, concentrating on the effects of food and oxygen deprivation. Rats have since been used to answer a wide range of basic science questions related to common human diseases in the fields of physiology, immunology, pharmacology, toxicology, nutrition, behavior and learning.
What are the advantages of using rats in human disease research?
Rats are an ideal choice for many labs due to their physiological similarity to humans. Also, since rats are easier to feed and smaller in size than other complex mammals, such as primates, they are a more convenient option for many researchers.
With the sequencing of the Brown Norway rat in 2004 by the Rat Genome Sequencing Project Consortium, led by the Baylor College of Medicine, it is now known that almost all disease-linked human genes have counterparts in the rat. Also, after advances in gene targeting and gene editing, it is possible to specifically modify the rat genome to produce knockouts and knockins, further enhancing our understanding of human disease through the development of powerful customized CRISPR rat models. Gene editing technologies allow scientists to use rats more often in their research. Better rat models are likely to decrease drug failure in clinical trials – currently standing at about 90% – which will decrease development costs and time to market. The sequenced genome will also unveil new targets for drug intervention.
What are the disadvantages of not using a rat model for certain types of studies?
In terms of the diseases that they are used to study, rats are usually not the model of choice when it comes to disorders related to neoplasms, blood and immune function. However, they’re more widely used in studying cardiovascular and metabolic disorders, as well as the onset of conditions that relate to the brain, digestive system, and various health problems related to behavior. Researchers are often advised to begin their studies using rat models in these areas, not only because they are a suitable model, but also due to the fact that rats have already helped scientists reach a few significant breakthroughs in these areas. Some of the disadvantages of choosing a different animal model for these fields could be difficulty in collecting accurate data, comparing data from previous studies that used rat models, and drawing definitive conclusions that could apply to future studies.
Researchers choose rats over other species for a number of reasons:
- The larger size when compared to mice makes handling, sampling and performing procedures easier.
- Some results suggest that the rat more accurately reflects human physiology than other species, mimicking human disease more accurately in many cases.
Behavioural research shows rats are more suited to studies on learning and cognition because they are more capable of learning tasks than other species.
- The rat’s ability to learn, remember, and interact gives them an advantage over other species, allowing for insights into the basic principles behind learning and behavior which can then be translated to human behavior and learning.
- It is now possible to more easily manipulate the rat genome to produce specific gene knockouts and knockins and apply results to our understanding of human genetic disorders.

### The power of rodent models
The rat had historically been the preferred animal model for biomedical research due to genetic and physiological similarities to humans. In 2001 however with the development of targeted genetic manipulation in mouse embryonic stem (ES) cells, rats took somewhat of a back seat as the rodent of choice for specific gene targeting because this technology was not yet available in the rat. As a result, mice became the more prominent model organism due to the availability of genetically modified lines where a gene of interest can be activated or inactivated in specific cells.
In mice, ES cells can be maintained in their pluripotent state easily. The same methods that allow this to be so in mice have failed in rats. Rat ES cells are very unstable, with any disturbance leading them to activate, so that they could not be implanted in an embryo and brought to term. The power of mouse models proved to be so clear however that the same abilities in rat continued to be desired and pursued.
Now, researchers no longer have to choose between the anatomical advantages of rats and the genetic tractability of mice, due to new methods for introducing genetic modifications into the rat genome. Significant progress was made in 2008 by Professor Austin Smith’s lab at Cambridge, and Professor Qi-Long Ying’s lab at the University of Southern California with the first targeting of rat ES cells achieved. Although the ability to target rat ES cells was then available, doing so was still difficult. It was around this time that gene editing came into being, specifically Zn Finger nucleases and TALENS. Although expensive to produce and not always precise, this became the preferred method to target rats in a specific manner.
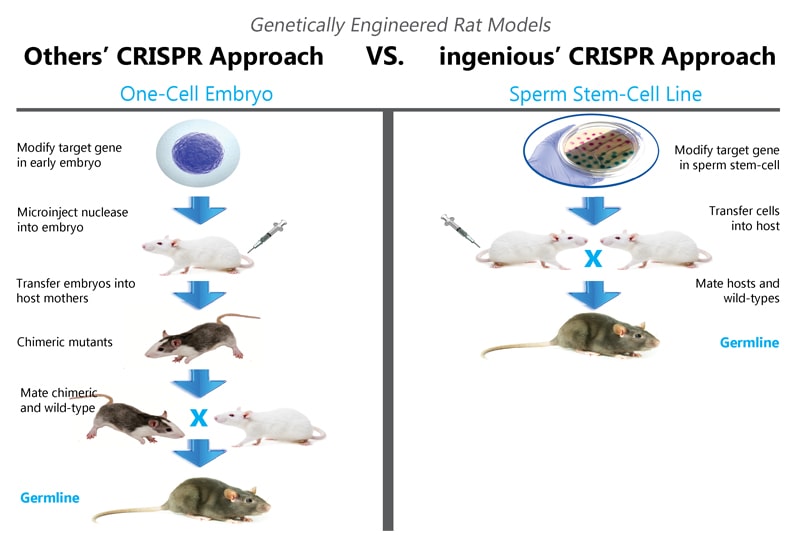
Then in 2012, CRISPR/Cas9 changed genome manipulation as we know it. With a much less expensive and more precise method for targeting genomes via embryos, the potential for rats as a model system of human disease could be pursued even easier. And more precise technologies have become available as well, such as using CRISPR to targeting rat spermatogonial stem cells. This alleviates the mosaicism common with targeting embryos, allowing for the production of specifically targeted germline rats in as little as 3 months. Researchers can now design experiments around the biological questions rather than the limitations of their animal model.
### Advancing cerebral palsy research with rat models
One area where rats have contributed to major insights is the field of cerebral palsy research. The term “cerebral palsy” denotes a highly variable syndrome affecting up to 3 out of every 1000 children born. It’s characterized primarily by motor impairment, but there are also a range of accompanying conditions such as seizure disorders and cognitive defects. In the process of finding a cure, the development of medicines to treat cerebral palsy is hampered by incomplete understanding of the condition’s earliest stages, when the brain first responds to an injury. Rodent models of cerebral palsy have been studied for almost forty years to fill the gaps in our knowledge, hopefully pointing the way to future treatments.
“ingenious was very competitively priced compared to other reputable transgenic companies. Most importantly, however, from the very beginning their knowledge and competence convinced me they would deliver on our project, so I trusted them and they fully met our expectations. Why use anyone else!”

Murray Clarke, PhDUniversity of Cambridge
Interested in using rats for your next research project? Click the button below to get a quote!
Why are rats used to model hypoxia-ischemia?
This research has almost exclusively used a single procedure to induce the condition in animal models. The hypoxia-ischemia model, first published in 1981, has brain damage similar to that observed in cerebral palsy patients. Originally the method was used on rat pups seven days after birth, when their brains are most similar to those of newborn babies. It has since been applied to a variety of species, to leverage similarities between the brains of humans and different animals. In recent years mice have been used with increasing frequency, to take advantage of genetically modified lines as well as the relatively lower costs compared with rats. However mice are less likely to survive the hypoxia-ischemia procedure, display a wider variety of brain damage, and have dramatic strain-to-strain differences. Rat models are therefore still widely used. Most studies focus on either testing how a specific genetic pathway responds to the brain injury, or how administering a drug affects the severity of damage after injury. These decades of work will enable the integration of new techniques and technologies for cerebral palsy research and maximize the utility of the hypoxia-ischemia model.
Citations and Resources
Colver A, Fairhurst C, Pharoah PO. 2014. Cerebral palsy. Lancet 383(9924): 1240-9.
Rumajogee P, Bregman T, Miller SP, Yager JY, Fehlings MG. 2016. Rodent Hypoxia-Ischemia Models for Cerebral Palsy Research: A Systematic Review. Front Neurol 7: 57.
Rice JE 3rd, Vannucci RC, Brierley JB. 1981. The influence of immaturity on hypoxic-ischemic brain damage in the rat. Ann Neurol 9(2): 131-41.